|
10/2/2023 0 Comments Benchling vs snapgene  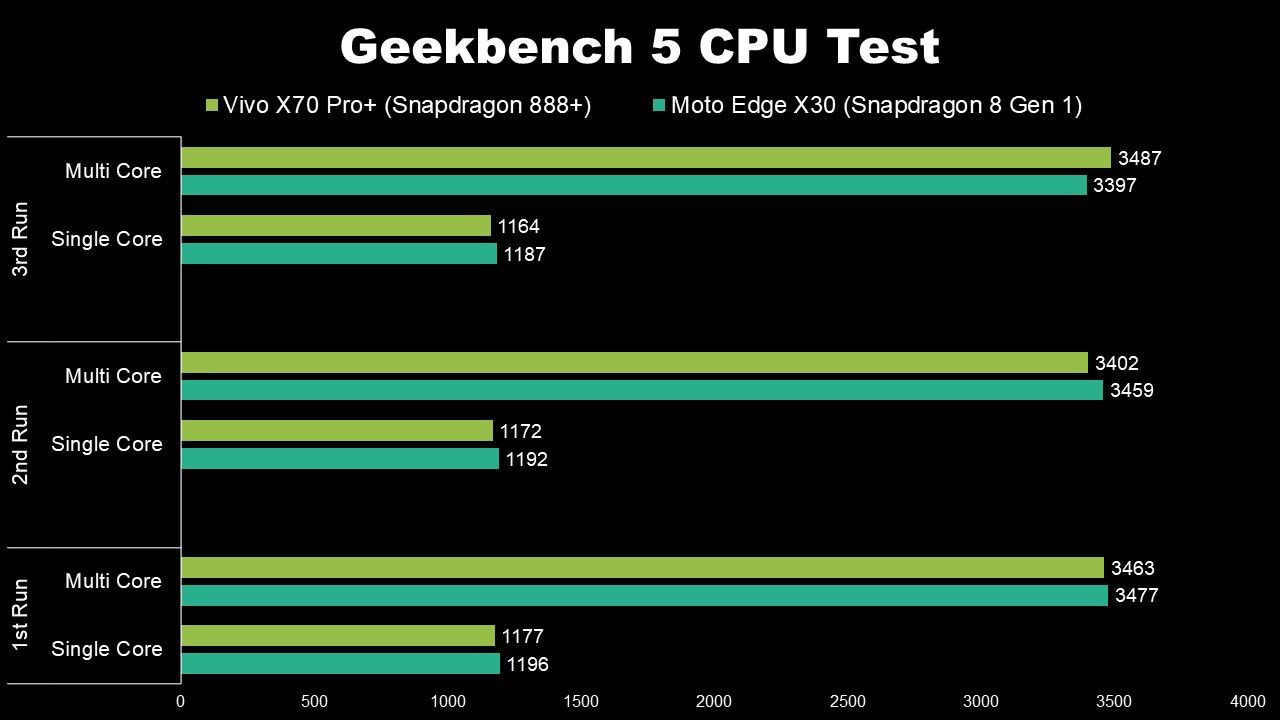
Basic Gibson Assembly can introduce errors at fragment junctions.Gibson Assembly Master Mixes are relatively expensive commercial products but are available from multiple sources.The difference in speed is magnified when using Gibson assembly to clone multiple fragments at one time. Finally, the technique is fast compared to traditional restriction enzyme cloning.Gibson assembly is a one-pot assembly technique for as many as 15 separate fragments.NON-restriction and ligation cloning technology allow the direct fusion of desired fragments.The use of PCR means that reliance on conveniently located restriction enzymes is eliminated.Gibson Assembly Pros & Cons Gibson Assembly ProsĪs one of several synthetic biology assembly techniques, Gibson assembly is a sequence-independent and seamless cloning technique. In this case, you can either band purify the resulting product or eliminate uncut template plasmid with the restriction enzyme DpnI. Alternatively, you can use inverse PCR to prepare your linear vector. You can eliminate this by band purifying your cut vector. When using restriction enzymes to linearize your vector, uncut plasmid can result in background colonies. To improve cloning efficiency, you can extend the reaction time to greater than one hour. Reactions involving 4 or more fragments and/or exceptionally long fragments should be incubated for one hour or longer. Simple assembly reactions are complete in 15 minutes. Gibson assembly results in covalently sealed molecules in vitro. Consult specific manufacturers for their guidelines. Suggested molar ratios of fragments to inserts are dependent on the size of the fragments and how many fragments are being assembled. Determine the concentration with UV spectroscopy. If additional bands are present, you should purify your insert from an agarose gel. If only your expected PCR product is present, you can purify the PCR product away from primers, dNTP’s and residual enzyme with a PCR clean-up column. QuantitationĪfter amplification, check your PCR products on an agarose gel. The overlapping regions selection box from SnapGene. When designing your cloning project, you can imagine that your primers have two distinct components, the target-specific primer for amplification and the 5’ tail that will create the overlap between the vector or adjacent fragments. 
The length of the homologous ends depends on the number of fragments being assembled and their length (see below). PCR primers need to be designed to amplify your fragment of interest and include 20 or more base pairs of homology to the vector or adjacent fragments in your cloned product. ► Learn to simulate Gibson Assembly in SnapGene Insert Preparation You have the option to prepare your vector by linearizing your plasmid with restriction enzymes or by using Reverse PCR. Insert fragments are always prepared by PCR. To perform Gibson assembly, you will need to prepare one or more inserts and your vector. This article refers to all these reactions generically as Gibson Assembly. Second-generation assembly reactions such as HiFi Assembly and Gibson Assembly Ultra improve on these limitations. The classic Gibson Assembly reaction can result in errors at fragment junctions and does not work with single-stranded fragments or short fragments. The reaction is designed to occur at 50☌, with all enzymes selected for their stability and activity at this temperature. T5 exonuclease chews back the 5’ end of double-stranded DNA to expose engineered overlaps. Polymerase begins filling in at the overhangs preventing excessive enzyme chewing. When stable compatible overhangs anneal, ligase completes the fusion of the DNA fragments. Once ligated, the DNA is protected from the exonuclease. These enzymes work together to fuse overlapping DNA fragments. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
